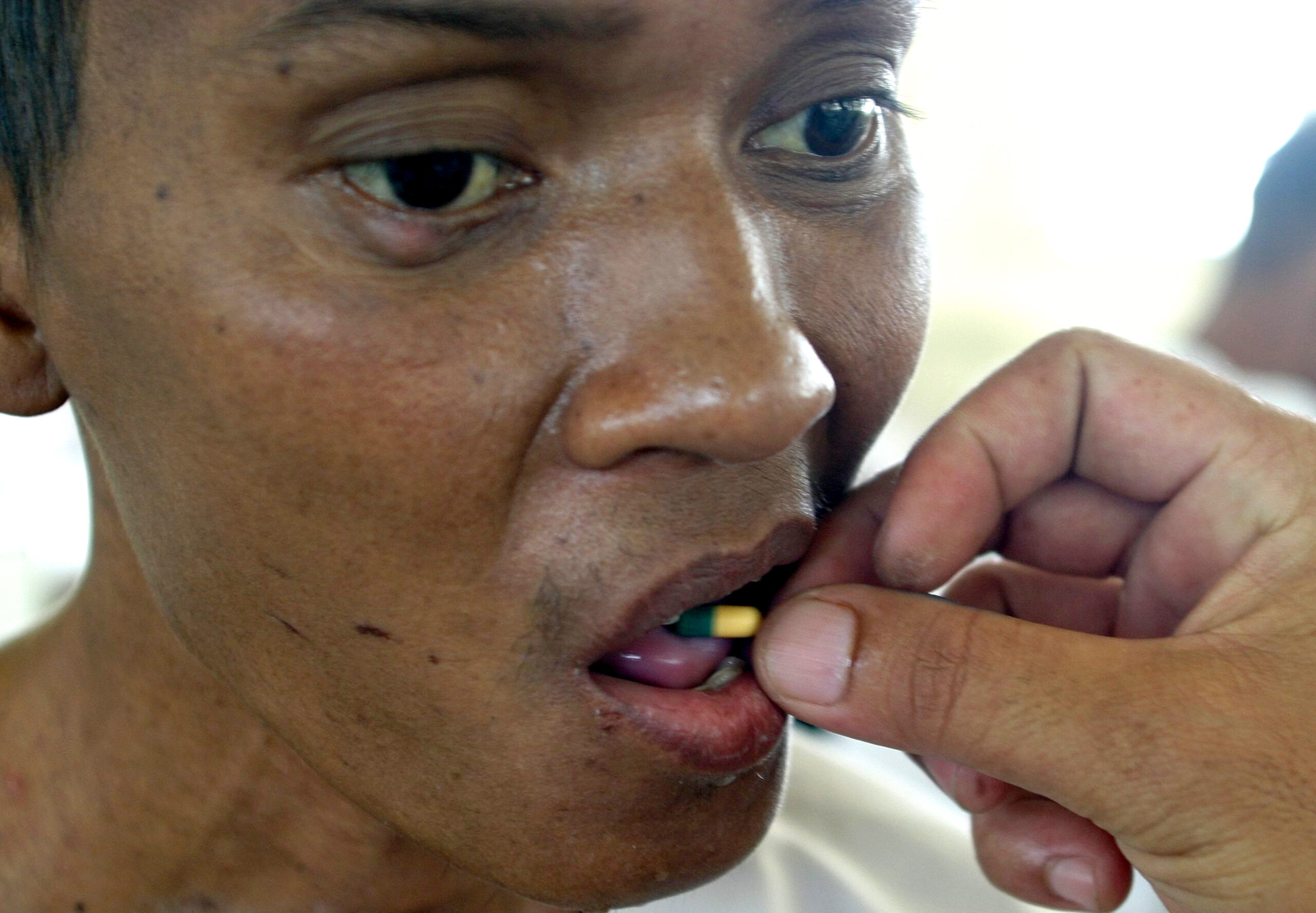
The pharmaceutical industry’s giants are testing their drugs on the poorest people of the poor countries, although they are destined to be sold to the rich. Why? Because in rich countries, there are hardly people who want to serve as human guinea pigs, while the poor from South Africa, India, Bulgaria, Zambia or Brazil are anxious to be so − in exchange for a small fee and some medical care. It does not matter if they put their health at risk and the new remedies, if approved, will be sold at prohibitive prices in the places where they live.
According to Sonia Shah*, who denounced the situation recently in Le Monde Diplomatique, GlaxoSmithKleine, in 2006, accomplished more than 50% of their clinical trials “outside the western markets” and, preferentially, “in low-cost countries.” On the race, there are also other potentates of the pharmaceutical industry, like Pfizer, Boehringer Ingelheim or AstraZeneca. The author comments: “The aim of the laboratories continues to be the conception of products destined for the regular customers of the western rich countries, whose health problems are associated with aging − cardiovascular illnesses, arthritis, hypertension and osteoporosis.” In other words, sicknesses that have nothing to do with the typical illnesses of the poor countries, where malaria, intestinal diseases or AIDS continue to kill millions, above all women and children. Why? Because the multinational companies are only interested in markets that produce, at least, 200 million dollars.
But developing countries suffer also from illnesses that affect the rich and since people from the former “offer” their bodies for the trials, at least they should be given access to those medicines at affordable prices. “According to the World Health Organization,” writes Shah, “80% of deaths provoked by chronic and non-contagious diseases occur in developing countries. For example, more than 90% of the world cases of Type 2 diabetes are in India and China. In certain regions of the African continent, one in five people is diabetic, and 20 million Africans suffer from hypertension.”
NOT IN MY BODY!
“The United States, with their average of ten annual prescriptions per inhabitant, constitutes the world’s largest pharmaceutical market, where the business volume increases 15% a year,” writes Shah. The industry, therefore, needs human guinea pigs − desperatly − “but the North Americans who accept to participate in therapeutic tests constitute only 5%.” To test a single medicine, which may not even become a reality because of its inefficiency, toxicity or secondary effects, needs a lot of people, at the lowest cost. “To be approved, all the drugs have to be tested in more than 4,000 patients. To choose them, previous examinations have to be carried out in more than 100,000 people!” she adds.
“Raw material” is abundant, cheap and credulous in all the countries where the poor abound like the Philippines, Malaysia, Thailand, Mexico, Estonia, Romania or Croatia. And as speed is also part of the business, great pharmaceutical companies make use of the so-called Contract Research Organizations (CROs) that take charge of the clinical trials abroad. The author exemplifies: Only “Covance brags of being able to make trials in 25,000 different centers, in a dozen countries. The specialized press is effervescent: ‘Practice skiing where there is snow, perform trials where there are illnesses,’ advertises one of the CROs.”
Supporters of this state of things proclaim the mutual advantages of the deslocalization of the tests. But, with the most limpid logic, the author reminds: “For a person to offer his own body to science is not quite the same as to use everyday the time clock at the factory.
Nothing guarantees that a participant will have better health at the end of the experiment. Such uncertainty and the risk of negative effects are, in fact, the first cause for the realization of the clinical trials.”
* Canadian journalist Sonia Shah is also author of the book The Body Hunters: Testing New Drugs on the World’s Poorest Patients (The New Press, New York, 2006). To know more, visit: www.soniashah.com